We’re starting off 2024 with a bang!! Today we’re hosting another expert Roundtable discussion and we’re joined by internationally recognized experts in the field. We’ll tackle everything from teaching about sepsis, to how to incorporate guidelines into education and practice, to future research directions in the field. This is a can’t-miss discussion. Let us know what you think and other sepsis questions you have!

Meet Our Guests
Dr. Derek Angus is a Professor at the University of Pittsburgh where he holds the Mitchell P. Fink Endowed Chair in Critical Care Medicine and is the Chair of the Department of Critical Care Medicine. He is a world-renowned researcher in a range of critical care topics including sepsis, has hundreds of publications, and has led numerous NIH-funded studies.
Dr. Hallie Prescott is an Associate Professor in Pulmonary and Critical Care Medicine at the University of Michigan. She is the Co-Chair of the Surviving Sepsis Campaign Guidelines and is also an internationally recognized expert due to her research in improving sepsis outcomes. She has been recognized by both medical journals and professional societies for her outstanding contributions to the field.
Summary of Episode Discussion Topics
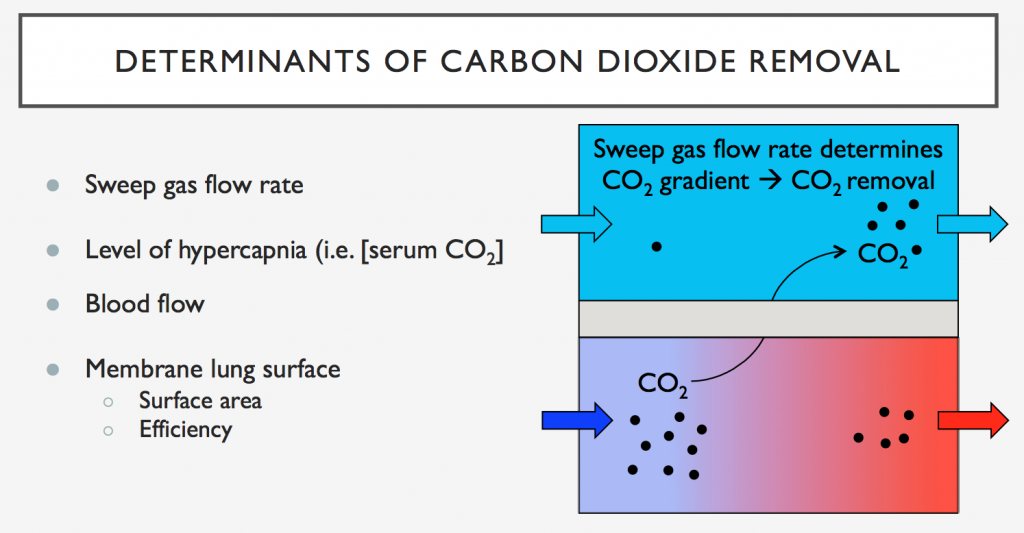
1. Sepsis Guidelines and Education
- Surviving Sepsis Guidelines: Stressed as essential reading for professionals in pulmonary and critical care. They provide a structured approach to sepsis management.
- Teaching Approaches: Transition from during-rounds teaching to focused, separate teaching sessions for trainees. Emphasizes the need to go beyond guidelines to include discussions on seminal articles, management strategies, and areas lacking robust data.
2. Clinical Skills and Decision Making in Sepsis Care
- Early Recognition and Polypharmacy: Highlighted the need for timely sepsis identification and caution against excessive polypharmacy.
- Mental Models in Care: Encourages building comprehensive mental models for understanding sepsis, stressing the importance of not just treating symptoms but understanding underlying causes.
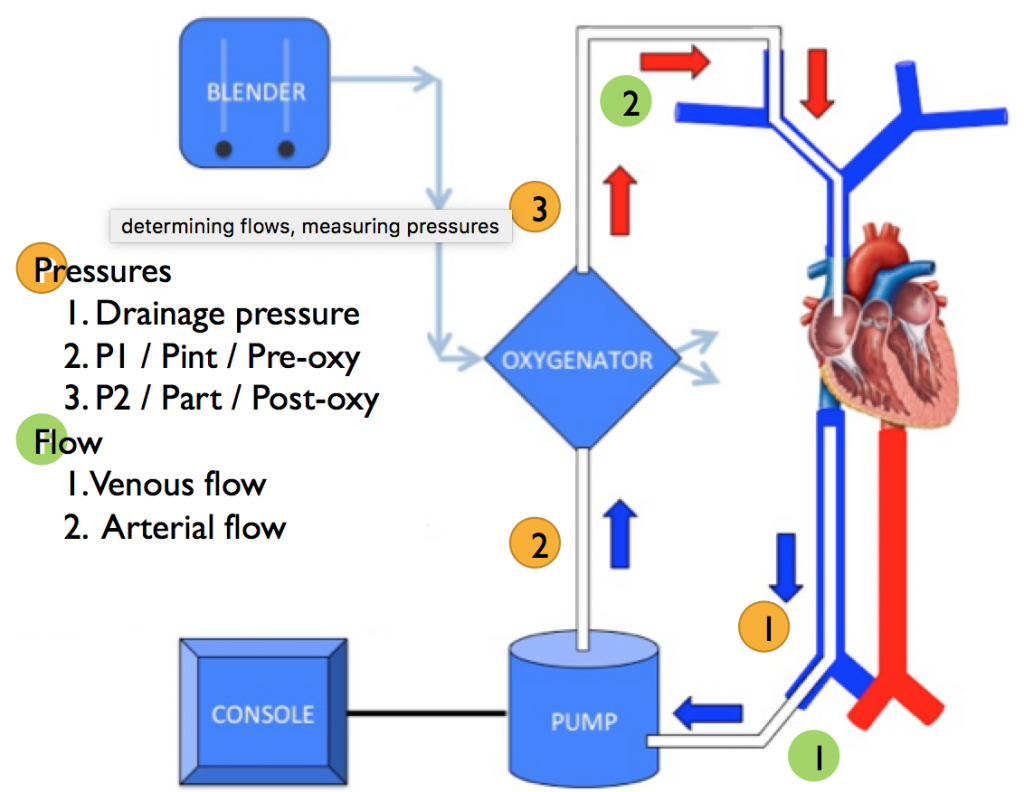
3. Implementation of Sepsis Guidelines
- Guideline Application in Bedside Care: Discusses the challenge of applying guidelines while considering patient-specific factors.
- Fluid Resuscitation Practices: Identifies fluid resuscitation as a key area for improvement, with a shift towards more conservative approaches.
- Overcoming Institutional Barriers: Addresses the fear of causing harm as a significant barrier to guideline implementation and emphasizes the need for balanced decision-making.
4. Advances in Sepsis Care and Prevention
- Pre-Hospital Sepsis Management: Explores the role of early intervention in community settings and the potential of wearables for early detection.
- Paramedic Role in Early Antibiotic Administration: Underlines the importance of starting antibiotics in the ambulance for suspected sepsis cases.
5. Recovery and Post-Discharge Care
- Post-Discharge Initiatives: Focuses on improving handoffs from ICU to ward and from hospital to home. Highlights the importance of medication reconciliation and clear communication with primary care.
- Challenges in Continuity of Care: Discusses the need for clear documentation and communication during patient transitions to ensure continuity of care.
6. Future Directions in Sepsis Treatment and Research
- Phenotyping for Targeted Treatment: The potential of identifying patient subgroups through phenotyping for more effective, tailored treatments.
- Adaptive Trial Designs: Advocates for large-scale adaptive platform trials that can test multiple interventions across diverse patient populations.
7. Personal Involvements and Perspectives
- Experts’ Current Work: The panelists share their ongoing projects and research in sepsis care, reflecting a commitment to advancing the field through comprehensive and adaptive approaches.
References and Further Reading
- Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, Machado FR, Mcintyre L, Ostermann M, Prescott HC, Schorr C, Simpson S, Wiersinga WJ, Alshamsi F, Angus DC, Arabi Y, Azevedo L, Beale R, Beilman G, Belley-Cote E, Burry L, Cecconi M, Centofanti J, Coz Yataco A, De Waele J, Dellinger RP, Doi K, Du B, Estenssoro E, Ferrer R, Gomersall C, Hodgson C, Hylander Møller M, Iwashyna T, Jacob S, Kleinpell R, Klompas M, Koh Y, Kumar A, Kwizera A, Lobo S, Masur H, McGloughlin S, Mehta S, Mehta Y, Mer M, Nunnally M, Oczkowski S, Osborn T, Papathanassoglou E, Perner A, Puskarich M, Roberts J, Schweickert W, Seckel M, Sevransky J, Sprung CL, Welte T, Zimmerman J, Levy M. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit Care Med. 2021 Nov 1;49(11):e1063-e1143. doi: 10.1097/CCM.0000000000005337. PMID: 34605781.
- Rudd KE, Kissoon N, Limmathurotsakul D, Bory S, Mutahunga B, Seymour CW, Angus DC, West TE. The global burden of sepsis: barriers and potential solutions. Crit Care. 2018 Sep 23;22(1):232. doi: 10.1186/s13054-018-2157-z. PMID: 30243300; PMCID: PMC6151187.
- Talisa VB, Yende S, Seymour CW, Angus DC. Arguing for Adaptive Clinical Trials in Sepsis. Front Immunol. 2018 Jun 28;9:1502. doi: 10.3389/fimmu.2018.01502. PMID: 30002660; PMCID: PMC6031704.
- Prescott HC, Angus DC. Enhancing Recovery From Sepsis: A Review. JAMA. 2018 Jan 2;319(1):62-75. doi: 10.1001/jama.2017.17687. PMID: 29297082; PMCID: PMC5839473.
- https://mi-hms.org/quality-initiatives/sepsis-initiative
- Kowalkowski M, Chou SH, McWilliams A, Lashley C, Murphy S, Rossman W, Papali A, Heffner A, Russo M, Burke L, Gibbs M, Taylor SP; Atrium Health ACORN Investigators. Structured, proactive care coordination versus usual care for Improving Morbidity during Post-Acute Care Transitions for Sepsis (IMPACTS): a pragmatic, randomized controlled trial. Trials. 2019 Nov 29;20(1):660. doi: 10.1186/s13063-019-3792-7. PMID: 31783900; PMCID: PMC6884908.
- Schmidt K, Worrack S, Von Korff M, Davydow D, Brunkhorst F, Ehlert U, Pausch C, Mehlhorn J, Schneider N, Scherag A, Freytag A, Reinhart K, Wensing M, Gensichen J; SMOOTH Study Group. Effect of a Primary Care Management Intervention on Mental Health-Related Quality of Life Among Survivors of Sepsis: A Randomized Clinical Trial. JAMA. 2016 Jun 28;315(24):2703-11. doi: 10.1001/jama.2016.7207. PMID: 27367877; PMCID: PMC5122319.
Podcast: Play in new window | Download
Subscribe: Apple Podcasts | Spotify | Amazon Music | Android | iHeartRadio | Podcast Index | RSS